Atomic Structure of Magnets
Claimed by Logan Vaupel (Fall 2016)
Magnets generate their magnetic fields at the atomic level.
The Main Idea
The magnetic field produced by a magnet is the sum of the magnetic dipole moments generated by each individual atom. These very small magnetic fields are generated much like those of circular current loops; however instead of being generated by electrons or charges flowing through a wire, the field in each individual atom is produced in one three different ways:
- An electron orbiting around the atomic nucleus. (See Figure 1)
- An electron rotating around its axis. (See Figure 1)
- The rotation of protons and neutrons within the nucleus of the atom.
All three of these situations produce a magnetic dipole proportional to the angular momentum. Together, the magnetic dipoles of all the atoms in the magnet sum to give the total magnetic dipole of the magnet. The magnetic field at an observation location can then be found from this dipole.
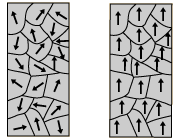
Although all atoms have electrons orbiting their nuclei, most materials are not magnetic. Each atom in these materials has a small magnetic dipole, however these dipoles are unaligned and disordered and therefore usually sum to zero. In magnetic materials, regions of magnetic dipoles line up. Although some of these regions cancel other regions out, enough regions align to produce a nonzero magnet field. This is allowed by interactions between atoms in certain elements (usually iron, nickel, cobalt, or alloys of these metals).
A Mathematical Model
We can model the magnetic dipole moment of a magnet by comparing the movement of electrons about the nucleus to the movement of current in a loop. In a current loop [math]\displaystyle{ {\mu = I \pi R^2} }[/math] when [math]\displaystyle{ R }[/math] is the radius of the loop. Since the units of [math]\displaystyle{ I }[/math] are [math]\displaystyle{ \frac{charge}{time} }[/math], the charge of an electron is [math]\displaystyle{ -e }[/math], and the period for one orbit around the nuclues is [math]\displaystyle{ t = \frac{2 \pi R}{v} }[/math] where [math]\displaystyle{ v }[/math] is the speed of the the electron, the magnetic dipole for one atom in a magnet simplifies to [math]\displaystyle{ \mu = \frac{e R v}{2} }[/math] where [math]\displaystyle{ R }[/math] is now the radius of the orbit.
The magnetic dipole is proportional to the angular momentum, [math]\displaystyle{ L }[/math] of the electron orbiting the nucleus. Assuming a circular orbit and assuming the speed of the electron is much less than the speed of light, [math]\displaystyle{ L = R p = R m v }[/math]. Multiplying the magnetic dipole by [math]\displaystyle{ \frac{m}{m} }[/math] reveals the proportionality of magnetic dipole and angular momentum [math]\displaystyle{ \mu = \frac{m}{m} \frac{e R v}{2} \Rightarrow \mu = \frac{e}{2 m} (R m v) = \frac{e}{2 m} L }[/math] From this equation the charge and mass of an electron and a proton can be plugged in to be compared. Because an electron weighs so much less than a proton, the magnetic dipole from the orbit of an electron is [math]\displaystyle{ 10^4 }[/math] times bigger than the magnetic dipole from the rotation of a proton or neutron in the nucleus, allowing the contributions from the protons and neutrons to be neglected.
For the purpose of calculating the magnetic dipole, it can be assumed that [math]\displaystyle{ L }[/math] is equal to Planck's Constant, [math]\displaystyle{ \hbar = 1.05 \times 10^{-34} \, J \cdot s }[/math]. Plugging in the charge and mass of an electron gives [math]\displaystyle{ \mu \approx 1 \times 10^{-23} \, A \cdot m^2 \, per \, atom }[/math].
Finally, if [math]\displaystyle{ \mu = N \mu_{atom} }[/math] where [math]\displaystyle{ N }[/math] is the number of atoms, this [math]\displaystyle{ \mu }[/math] is the magnetic dipole of the magnet.
However, it is worth noting that we made several simplifying assumptions to create this model. First, we assumed that the movement of electrons in their orbitals were moving in a circular path, creating angular momentum. In reality, atomic orbitals are much more complex probability distributions for finding an electron at a specific location. The different energy levels of electron orbits form different shapes with different angular momentums. The "s" orbital is a sphere with a net zero angular momentum, while the "p, d, and f" orbitals are asymmetrical and thus produce an angular momentum, which can produce a magnetic dipole moment. Also, we assumed that there was only one unpaired electron moving within its orbit per atom, when in reality, many materials, such as iron, have two or more unpaired electrons that contribute to the net magnetic dipole moment. A more accurate model would also take into account not just the magnetic contribution of individual atoms, but also how they impact one another. We also assumed that the atoms were perfectly aligned, when this would most certainly never happen in reality. Also, a deeper understanding of the math and physics behind why certain materials are magnetic while others are not requires knowledge of quantum mechanics.. Thus, a more accurate model would produce far more complex calculations. Nevertheless, this simpler model still gives useful insight into how the movement of electrons within atomic orbitals within a material can create a macro magnetic dipole effect.
A Computational Model
Here is a link that to glowscript showing a model of an electron orbiting a nucleus, which is how the magnetic dipole in a magnet is formed.
Examples
Simple
A bar magnet made from iron has a mass of 72 g. What is the magnetic dipole of the bar magnet?
[math]\displaystyle{ N = \left (\frac{72 \, g \, iron}{56 \, \frac{g \, iron}{mol \, iron}} \right ) \left (6.022 \times 10^{23} \frac{atoms}{mol} \right ) = 7.74 \times 10^{23} \, atoms }[/math]
[math]\displaystyle{ \mu = (7.74 \times 10^{23} \, atoms) \left (1 \times 10^{-23} \, \frac{A \cdot m^2}{atom} \right ) = 7.74 \, A \cdot m^2 }[/math]
Middling
A bar magnet that is 15% mass iron and 85% mass nickel has a mass of 126 g. What is the magnetic dipole of the bar magnet?
[math]\displaystyle{ 126 \, g \cdot 0.15 = 18.9 \, g \, iron }[/math]
[math]\displaystyle{ 126 \, g \cdot 0.85 = 107.1 \, g \, nickel }[/math]
[math]\displaystyle{ N = \left (\frac{18.9 \, g \, iron}{56 \, \frac{g \, iron}{mol \, iron}} \right ) \left (6.022 \times 10^{23} \frac{atoms}{mol} \right ) = 2.03 \times 10^{23} \, atoms }[/math]
[math]\displaystyle{ N = \left (\frac{107.1 \, g \, nickel}{59 \, \frac{g \, nickel}{mol \, iron}} \right ) \left (6.022 \times 10^{23} \frac{atoms}{mol} \right ) = 1.09 \times 10^{24} \, atoms }[/math]
[math]\displaystyle{ 2.03 \times 10^{23} \, atoms + 1.09 \times 10^{24} \, atoms = 1.29 \times 10^{24} \, atoms }[/math]
[math]\displaystyle{ \mu = (1.29 \times 10^{24} \, atoms) \left (1 \times 10^{-23} \, \frac{A \cdot m^2}{atom} \right ) = 12.9 \, A \cdot m^2 }[/math]
Difficult
A compass originally points north. A bar magnet made of iron is placed [math]\displaystyle{ 20 \, cm }[/math] west of the compass on axis, with the north end of the magnet pointing towards the compass. The compass deflects [math]\displaystyle{ 55^\circ }[/math]. What is the mass of the magnet?
[math]\displaystyle{ \overrightarrow{B}_{net} = \overrightarrow{B}_{Earth} + \overrightarrow{B}_{magnet} }[/math]
[math]\displaystyle{ B_{magnet} = (2 \times 10^{-5} \,T) \tan{55^\circ} }[/math]
[math]\displaystyle{ B_{magnet} = 2.86 \times 10^{-5} \,T }[/math]
[math]\displaystyle{ B_{magnet} = \frac{\mu_0}{4 \pi} \frac{2 \mu}{r^3} }[/math]
[math]\displaystyle{ \mu = \frac{r^3 B_{magnet}}{2} \frac{4 \pi}{\mu_0} }[/math]
[math]\displaystyle{ \mu = \frac{(0.20 \, m)^3 (2.86 \times 10^{-5} \,T)}{2} \left (1 \times 10^7 \, T \cdot \frac{m}{A} \right) }[/math]
[math]\displaystyle{ \mu = 1.14 \, A \cdot m^2 }[/math]
[math]\displaystyle{ number \, of \, atoms = \frac{1.14 \, A \cdot m^2}{1 \times 10^{-23} \, \frac{A \cdot m^2}{atom}} }[/math]
[math]\displaystyle{ number \, of \, atoms = 1.14 \times 10^{23} \, atoms }[/math]
[math]\displaystyle{ mass \, of \, bar = (1.14 \times 10^{23} \, atoms) \left ( \frac{56 \, \frac{g \, iron}{mol \, iron}}{6.022 \times 10^{23} \frac{atoms}{mol}} \right ) }[/math]
[math]\displaystyle{ mass \, of \, bar = 10.6 \, g \, iron }[/math]
Connectedness
- How is this topic connected to something that you are interested in?
- I've always thought magnets were interesting since I was a child. I have always been fascinated by magnetic force because you can't see it working and as a child it always seemed like magic!
- How is it connected to your major?
- Chemical Engineers don't deal with E&M very much at all however Physics II is required for graduation.
- Is there an interesting industrial application?
- As the atomic structure of magnets is mostly just concept, there aren't really an industrial applications aside from the production of bar magnets.
History
The "first" magnets were called lodestones and are just naturally magnetic pieces of metal. There are records of lodestones dating up to 2500 years ago from all over the world. By the 12th century, humans discovered they could suspend small pieces of lodestones which would allow them to rotate, and then used them for navigation. We still use compasses to this day!
See also
The Angular Momentum Principle
Further reading
External links
References
Chabay, R., & Sherwood, B. (2015). Magnetic Field. In Matter & interactions (4th ed., Vol. 2, pp. 693-698). Hoboken, NJ: Wiley.
Ferromagnetism. (n.d.). Retrieved December 5, 2015, from http://hyperphysics.phy-astr.gsu.edu/hbase/solids/ferro.html#c4
Lodestone. (n.d.). Retrieved December 5, 2015, from https://en.wikipedia.org/wiki/Lodestone
Origin of Magnetism. (n.d.). Retrieved December 5, 2015, from http://www.gitam.edu/eresource/Engg_Phys/semester_2/magnetic/intro.htm