Quantum Tunneling through Potential Barriers: Difference between revisions
No edit summary |
|||
| (7 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
'''Page Claimed By: Haripratiik Arunkumar Malarkodi Spring | '''<big><big>Page Claimed By: Haripratiik Arunkumar Malarkodi, Spring 2026</big></big>''' | ||
'''Quantum tunneling''' is a | '''Quantum tunneling''' is a quantum mechanics effect where a particle can be found on the other side of a potential barrier even when, classically, it does not have enough energy to cross it. This sounds impossible from a classical point of view, but it makes sense once the particle is treated as a wavefunction instead of as a tiny ball following one exact path. | ||
The main idea is that the wavefunction does not suddenly become zero when it reaches a finite barrier. Instead, it can extend into the barrier and decay as it moves through it. If the barrier is thin enough, some of the wavefunction can still exist on the far side. That gives a nonzero probability of finding the particle past the barrier. | |||
This matters because tunneling is not just a strange theoretical effect. It helps explain alpha decay, fusion in stars, scanning tunneling microscopes, and tunneling effects in semiconductor devices. | |||
==Overview== | ==Overview== | ||
A simple way to picture tunneling is to compare a quantum particle to a ball rolling toward a hill. In classical mechanics, if the ball does not have enough energy to get over the hill, it rolls back. There is no chance that it appears on the other side. | |||
A | Quantum mechanics gives a different result. A particle is described by a wavefunction, and the wavefunction can enter regions that would be forbidden in classical mechanics. For a rectangular barrier of height <math>V_0</math> and width <math>L</math>, a particle with energy <math>E<V_0</math> cannot cross the barrier classically. However, its wavefunction can decay inside the barrier. If the barrier is not too wide, part of the wavefunction reaches the other side and becomes a transmitted wave. | ||
The important point is that tunneling is probabilistic. We cannot say exactly when one particle will tunnel, but we can calculate the probability that it will. | |||
[[File:Quantum-tunneling.png|thumb|center|500px|Quantum tunneling through a square potential barrier. The diagram shows an incoming wave encountering a finite barrier and a smaller transmitted wave appearing on the far side. Image by MikeRun, licensed under Creative Commons Attribution-ShareAlike 4.0 International, via Wikimedia Commons.]] | |||
===Classical Expectations=== | ===Classical Expectations=== | ||
| Line 17: | Line 21: | ||
Under classical mechanics, a particle can cross a potential barrier only if its total energy is greater than the barrier's potential energy. Therefore: | Under classical mechanics, a particle can cross a potential barrier only if its total energy is greater than the barrier's potential energy. Therefore: | ||
* If <math>E > V_0</math>, the particle can pass over the barrier. | * If <math>E>V_0</math>, the particle can pass over the barrier. | ||
* If <math>E < V_0</math>, the particle cannot pass through or over the barrier. | * If <math>E<V_0</math>, the particle cannot pass through or over the barrier. | ||
* The classical transmission probability for <math>E < V_0</math> is exactly zero. | * The classical transmission probability for <math>E<V_0</math> is exactly zero. | ||
This classical model works well for macroscopic objects, but it fails for microscopic particles such as electrons, protons, and alpha particles. | This classical model works well for macroscopic objects, but it fails for microscopic particles such as electrons, protons, and alpha particles. | ||
| Line 27: | Line 31: | ||
====Wave Behavior of Particles==== | ====Wave Behavior of Particles==== | ||
Quantum mechanics describes particles using wavefunctions. A wavefunction, usually written as <math>\psi(x)</math>, contains information about the probability of finding a particle at different positions. The probability density is | Quantum mechanics describes particles using wavefunctions. A wavefunction, usually written as <math>\psi(x)</math>, contains information about the probability of finding a particle at different positions. The probability density is | ||
<math>|\psi(x)|^2</math>. | <math>|\psi(x)|^2</math>. | ||
| Line 37: | Line 41: | ||
====Important Intuition==== | ====Important Intuition==== | ||
The tunneling probability is very sensitive to the barrier. Three changes matter the most: | |||
* | * Increasing the barrier width <math>L</math> makes tunneling much less likely. | ||
* | * Increasing the barrier height <math>V_0</math> makes tunneling much less likely when the particle energy stays the same. | ||
* | * Increasing the particle mass <math>m</math> makes tunneling much less likely. | ||
This is why tunneling is | This is why tunneling is noticeable for very small particles, like electrons, but not for everyday objects. A baseball technically has a quantum wavefunction, but its mass is so large that the tunneling probability through an ordinary wall is effectively zero. | ||
====Heisenberg Uncertainty Principle==== | ====Heisenberg Uncertainty Principle==== | ||
The | The uncertainty principle is sometimes used to give intuition for tunneling, but it should not be treated as the main explanation. A common oversimplification is to say that the particle "borrows energy" for a short time. That picture can be helpful at first, but it is not how tunneling is actually calculated. | ||
The | The more reliable explanation comes from the Schrödinger equation. When the wavefunction meets a finite barrier, the boundary conditions force part of the wavefunction to extend into the barrier. Inside the barrier, the wavefunction decays exponentially instead of disappearing instantly. | ||
====The Schrödinger Equation==== | ====The Schrödinger Equation==== | ||
| Line 129: | Line 133: | ||
At <math>x=0</math>: | At <math>x=0</math>: | ||
<math> | <math>A+B=C+D</math> | ||
<math> | <math>ik(A-B)=-\kappa C+\kappa D</math> | ||
At <math>x=L</math>: | At <math>x=L</math>: | ||
<math>\ | <math>Ce^{-\kappa L}+De^{\kappa L}=Fe^{ikL}</math> | ||
<math>\ | <math>-\kappa Ce^{-\kappa L}+\kappa De^{\kappa L}=ikFe^{ikL}</math> | ||
These four equations can be solved for the unknown amplitudes <math>B</math>, <math>C</math>, <math>D</math>, and <math>F</math> in terms of the incoming amplitude <math>A</math>. | These four equations can be solved for the unknown amplitudes <math>B</math>, <math>C</math>, <math>D</math>, and <math>F</math> in terms of the incoming amplitude <math>A</math>. | ||
| Line 146: | Line 150: | ||
<math>T=\frac{\text{transmitted intensity}}{\text{incident intensity}}</math>. | <math>T=\frac{\text{transmitted intensity}}{\text{incident intensity}}</math>. | ||
Since intensity is proportional to the squared amplitude, | |||
<math>T=\frac{|F|^2}{|A|^2}</math>. | |||
For a rectangular barrier with <math>E<V_0</math>, the exact transmission coefficient is | For a rectangular barrier with <math>E<V_0</math>, the exact transmission coefficient is | ||
| Line 167: | Line 175: | ||
==Computational Model== | ==Computational Model== | ||
The | The model below is an original GlowScript/Trinket visualization of quantum tunneling through a rectangular potential barrier. It lets the user change the particle energy <math>E</math>, barrier height <math>V_0</math>, and barrier width <math>L</math>. The model shows the oscillating wave before the barrier, the decaying wave inside the barrier, and the smaller transmitted wave after the barrier. | ||
'''Original GlowScript/Trinket model:''' [https://trinket.io/embed/glowscript/caf10538764a?start=result Quantum Tunneling through a Rectangular Potential Barrier] | |||
<iframe src="https://trinket.io/embed/glowscript/caf10538764a?start=result" | |||
width="100%" | |||
height="500" | |||
frameborder="0" | |||
marginwidth="0" | |||
marginheight="0" | |||
allowfullscreen> | |||
</iframe> | |||
If the embedded model does not load inside the wiki, open the linked Trinket model above in a new tab. This model was created for this page and is meant to show the main qualitative idea: the wavefunction does not instantly become zero inside the barrier. It decays, and the amount left on the far side depends strongly on the barrier height, barrier width, and particle energy. | |||
The PhET quantum tunneling simulation below is also useful because it lets users compare plane waves and wave packets for different potential shapes. | |||
<iframe src="https://phet.colorado.edu/sims/cheerpj/quantum-tunneling/latest/quantum-tunneling.html?simulation=quantum-tunneling" | <iframe src="https://phet.colorado.edu/sims/cheerpj/quantum-tunneling/latest/quantum-tunneling.html?simulation=quantum-tunneling" | ||
width="800" | width="800" | ||
height="600" | height="600" | ||
frameborder="0" | |||
marginwidth="0" | |||
marginheight="0" | |||
allowfullscreen> | allowfullscreen> | ||
</iframe> | </iframe> | ||
When using the | When using the simulations, try the following: | ||
* Set the particle energy below the barrier height. | * Set the particle energy below the barrier height. | ||
| Line 184: | Line 210: | ||
==Common Exam Mistakes== | ==Common Exam Mistakes== | ||
* ''' | * '''Thinking tunneling is classically allowed.''' If <math>E<V_0</math>, classical mechanics predicts no transmission. Tunneling is a quantum effect. | ||
* '''Forgetting the | * '''Forgetting that the probability drops exponentially.''' The factor <math>e^{-2\kappa L}</math> means that even a small increase in barrier width can make tunneling much less likely. | ||
* ''' | * '''Mixing up <math>k</math> and <math>\kappa</math>.''' The symbol <math>k</math> describes the oscillating wave outside the barrier. The symbol <math>\kappa</math> describes the decaying wave inside the barrier. | ||
* ''' | * '''Saying the particle climbs over the barrier.''' In tunneling, the particle is not following a classical path over the barrier. The wavefunction extends through the barrier. | ||
* '''Ignoring mass.''' | * '''Ignoring mass.''' Since <math>\kappa</math> depends on mass, heavier particles tunnel much less easily than lighter particles. | ||
==Examples== | ==Examples== | ||
| Line 385: | Line 411: | ||
<math> | <math> | ||
L\approx8. | L\approx8.54\times10^{-10}\text{ m} | ||
</math>. | </math>. | ||
| Line 391: | Line 417: | ||
<math> | <math> | ||
\boxed{0. | \boxed{0.854\text{ nm}} | ||
</math>. | </math>. | ||
==Applications== | ==Applications== | ||
===Nuclear Fusion in Stars=== | |||
Quantum tunneling helps explain how nuclear fusion can occur inside stars. Protons repel each other because they are positively charged. Classically, many protons in a star's core would not have enough energy to overcome this electric repulsion. Quantum tunneling gives them a small probability of getting close enough for the strong nuclear force to bind them together. | Quantum tunneling helps explain how nuclear fusion can occur inside stars. Protons repel each other because they are positively charged. Classically, many protons in a star's core would not have enough energy to overcome this electric repulsion. Quantum tunneling gives them a small probability of getting close enough for the strong nuclear force to bind them together. | ||
===Alpha Decay=== | |||
Alpha decay occurs when an unstable nucleus emits an alpha particle. The alpha particle is trapped inside the nucleus by a potential barrier. Classically, it may not have enough energy to escape. Quantum mechanically, it can tunnel through the barrier and leave the nucleus. | Alpha decay occurs when an unstable nucleus emits an alpha particle. The alpha particle is trapped inside the nucleus by a potential barrier. Classically, it may not have enough energy to escape. Quantum mechanically, it can tunnel through the barrier and leave the nucleus. | ||
===Scanning Tunneling Microscopy=== | |||
A scanning tunneling microscope, or STM, uses tunneling current to image surfaces at the atomic scale. When a sharp conducting tip is brought very close to a surface, electrons can tunnel between the tip and the surface. The tunneling current is extremely sensitive to distance, which allows the STM to detect tiny changes in surface height. | A scanning tunneling microscope, or STM, uses tunneling current to image surfaces at the atomic scale. When a sharp conducting tip is brought very close to a surface, electrons can tunnel between the tip and the surface. The tunneling current is extremely sensitive to distance, which allows the STM to detect tiny changes in surface height. | ||
===Semiconductor Devices=== | |||
Some semiconductor devices rely on tunneling. Tunnel diodes, flash memory, and very small transistors can involve tunneling effects. As electronic devices become smaller, tunneling becomes more important because electrons interact with barriers only a few nanometers wide. | Some semiconductor devices rely on tunneling. Tunnel diodes, flash memory, and very small transistors can involve tunneling effects. As electronic devices become smaller, tunneling becomes more important because electrons interact with barriers only a few nanometers wide. | ||
| Line 414: | Line 440: | ||
==Connection to Matter and Interactions== | ==Connection to Matter and Interactions== | ||
In Matter and Interactions, | In Matter and Interactions, energy is often used to decide what motion is possible. For a classical particle, if <math>E<V_0</math>, the particle cannot cross the barrier. Quantum tunneling is interesting because it shows where that classical energy reasoning stops being complete. | ||
The particle still does not have enough classical energy to go over the barrier. The difference is that quantum mechanics describes the particle with a wavefunction. That wavefunction can extend into the barrier and sometimes reach the other side. So instead of asking only whether the particle has enough energy to cross, we ask what probability the wavefunction gives for finding the particle past the barrier. | |||
==Historical Background== | ==Historical Background== | ||
| Line 433: | Line 459: | ||
==External Resources== | ==External Resources== | ||
* [https://commons.wikimedia.org/wiki/File:Quantum-tunneling.svg Wikimedia Commons: Quantum-tunneling.svg] | |||
* [https://phet.colorado.edu/en/simulations/quantum-tunneling PhET: Quantum Tunneling and Wave Packets] | * [https://phet.colorado.edu/en/simulations/quantum-tunneling PhET: Quantum Tunneling and Wave Packets] | ||
* [https://openstax.org | * [https://openstax.org/books/university-physics-volume-3/pages/7-6-the-quantum-tunneling-of-particles-through-potential-barriers OpenStax: Quantum Tunneling of Particles through Potential Barriers] | ||
* [https://phys.libretexts.org/Bookshelves/University_Physics/University_Physics_%28OpenStax%29/University_Physics_III_-_Optics_and_Modern_Physics_%28OpenStax%29/07%3A_Quantum_Mechanics/7.07%3A_Quantum_Tunneling_of_Particles_through_Potential_Barriers LibreTexts: Quantum Tunneling of Particles through Potential Barriers] | * [https://phys.libretexts.org/Bookshelves/University_Physics/University_Physics_%28OpenStax%29/University_Physics_III_-_Optics_and_Modern_Physics_%28OpenStax%29/07%3A_Quantum_Mechanics/7.07%3A_Quantum_Tunneling_of_Particles_through_Potential_Barriers LibreTexts: Quantum Tunneling of Particles through Potential Barriers] | ||
* [http://hyperphysics.phy-astr.gsu.edu/hbase/quantum/barr.html HyperPhysics: Barrier Penetration] | * [http://hyperphysics.phy-astr.gsu.edu/hbase/quantum/barr.html HyperPhysics: Barrier Penetration] | ||
| Line 440: | Line 467: | ||
==References== | ==References== | ||
* OpenStax, ''University Physics Volume 3'', Chapter 7: Quantum Mechanics. | * MikeRun, ''Quantum-tunneling.svg'', Wikimedia Commons. Licensed under Creative Commons Attribution-ShareAlike 4.0 International. | ||
* OpenStax, ''University Physics Volume 3'', Chapter 7: Quantum Mechanics. Licensed under Creative Commons Attribution 4.0 International. | |||
* LibreTexts, ''Quantum Tunneling of Particles through Potential Barriers''. | * LibreTexts, ''Quantum Tunneling of Particles through Potential Barriers''. | ||
* HyperPhysics, ''Barrier Penetration''. | * HyperPhysics, ''Barrier Penetration''. | ||
* PhET Interactive Simulations, ''Quantum Tunneling and Wave Packets''. | * PhET Interactive Simulations, ''Quantum Tunneling and Wave Packets''. | ||
* Original GlowScript/Trinket model by Haripratiik Arunkumar Malarkodi, Spring 2026. | |||
[[Category: Quantum Mechanics]] | [[Category: Quantum Mechanics]] | ||
Latest revision as of 13:17, 27 April 2026
Page Claimed By: Haripratiik Arunkumar Malarkodi, Spring 2026
Quantum tunneling is a quantum mechanics effect where a particle can be found on the other side of a potential barrier even when, classically, it does not have enough energy to cross it. This sounds impossible from a classical point of view, but it makes sense once the particle is treated as a wavefunction instead of as a tiny ball following one exact path.
The main idea is that the wavefunction does not suddenly become zero when it reaches a finite barrier. Instead, it can extend into the barrier and decay as it moves through it. If the barrier is thin enough, some of the wavefunction can still exist on the far side. That gives a nonzero probability of finding the particle past the barrier.
This matters because tunneling is not just a strange theoretical effect. It helps explain alpha decay, fusion in stars, scanning tunneling microscopes, and tunneling effects in semiconductor devices.
Overview
A simple way to picture tunneling is to compare a quantum particle to a ball rolling toward a hill. In classical mechanics, if the ball does not have enough energy to get over the hill, it rolls back. There is no chance that it appears on the other side.
Quantum mechanics gives a different result. A particle is described by a wavefunction, and the wavefunction can enter regions that would be forbidden in classical mechanics. For a rectangular barrier of height [math]\displaystyle{ V_0 }[/math] and width [math]\displaystyle{ L }[/math], a particle with energy [math]\displaystyle{ E\lt V_0 }[/math] cannot cross the barrier classically. However, its wavefunction can decay inside the barrier. If the barrier is not too wide, part of the wavefunction reaches the other side and becomes a transmitted wave.
The important point is that tunneling is probabilistic. We cannot say exactly when one particle will tunnel, but we can calculate the probability that it will.
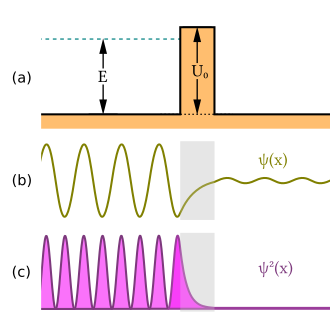
Classical Expectations
Under classical mechanics, a particle can cross a potential barrier only if its total energy is greater than the barrier's potential energy. Therefore:
- If [math]\displaystyle{ E\gt V_0 }[/math], the particle can pass over the barrier.
- If [math]\displaystyle{ E\lt V_0 }[/math], the particle cannot pass through or over the barrier.
- The classical transmission probability for [math]\displaystyle{ E\lt V_0 }[/math] is exactly zero.
This classical model works well for macroscopic objects, but it fails for microscopic particles such as electrons, protons, and alpha particles.
Quantum Theory
Wave Behavior of Particles
Quantum mechanics describes particles using wavefunctions. A wavefunction, usually written as [math]\displaystyle{ \psi(x) }[/math], contains information about the probability of finding a particle at different positions. The probability density is
[math]\displaystyle{ |\psi(x)|^2 }[/math].
When a quantum particle reaches a finite potential barrier, its wavefunction must satisfy boundary conditions at the edges of the barrier. Since waves generally do not suddenly disappear at a boundary, the wavefunction extends into the barrier. Inside the barrier, the wavefunction has an exponentially decaying form. If the barrier is thin enough, the wavefunction can still have a nonzero value on the far side.
This is the origin of tunneling.
Important Intuition
The tunneling probability is very sensitive to the barrier. Three changes matter the most:
- Increasing the barrier width [math]\displaystyle{ L }[/math] makes tunneling much less likely.
- Increasing the barrier height [math]\displaystyle{ V_0 }[/math] makes tunneling much less likely when the particle energy stays the same.
- Increasing the particle mass [math]\displaystyle{ m }[/math] makes tunneling much less likely.
This is why tunneling is noticeable for very small particles, like electrons, but not for everyday objects. A baseball technically has a quantum wavefunction, but its mass is so large that the tunneling probability through an ordinary wall is effectively zero.
Heisenberg Uncertainty Principle
The uncertainty principle is sometimes used to give intuition for tunneling, but it should not be treated as the main explanation. A common oversimplification is to say that the particle "borrows energy" for a short time. That picture can be helpful at first, but it is not how tunneling is actually calculated.
The more reliable explanation comes from the Schrödinger equation. When the wavefunction meets a finite barrier, the boundary conditions force part of the wavefunction to extend into the barrier. Inside the barrier, the wavefunction decays exponentially instead of disappearing instantly.
The Schrödinger Equation
The time-independent Schrödinger equation determines the allowed wavefunctions for a particle in a given potential energy function:
[math]\displaystyle{ -\frac{\hbar^2}{2m}\frac{d^2\psi(x)}{dx^2}+V(x)\psi(x)=E\psi(x) }[/math].
For a finite potential barrier, the wavefunction must be continuous at each boundary. Its derivative must also be continuous as long as the potential is finite. These conditions allow us to solve for the reflected and transmitted parts of the wave.
Mathematical Model
Consider a rectangular potential barrier:
[math]\displaystyle{ V(x)= \begin{cases} 0, & x\lt 0 \\ V_0, & 0\le x\le L \\ 0, & x\gt L \end{cases} }[/math]
Assume the particle energy satisfies
[math]\displaystyle{ 0\lt E\lt V_0 }[/math].
This means the particle does not have enough energy to cross the barrier classically.
Wave Equations
In each region, the Schrödinger equation has a different form.
Region I: Before the barrier, [math]\displaystyle{ x\lt 0 }[/math]
[math]\displaystyle{ -\frac{\hbar^2}{2m}\frac{d^2\psi_I}{dx^2}=E\psi_I }[/math]
The general solution is
[math]\displaystyle{ \psi_I(x)=Ae^{ikx}+Be^{-ikx} }[/math].
Here:
- [math]\displaystyle{ Ae^{ikx} }[/math] is the incident wave moving toward the barrier.
- [math]\displaystyle{ Be^{-ikx} }[/math] is the reflected wave moving away from the barrier.
Region II: Inside the barrier, [math]\displaystyle{ 0\le x\le L }[/math]
[math]\displaystyle{ -\frac{\hbar^2}{2m}\frac{d^2\psi_{II}}{dx^2}+V_0\psi_{II}=E\psi_{II} }[/math]
Since [math]\displaystyle{ E\lt V_0 }[/math], the solution is exponential rather than sinusoidal:
[math]\displaystyle{ \psi_{II}(x)=Ce^{-\kappa x}+De^{\kappa x} }[/math].
This shows that the wavefunction decays inside the barrier instead of oscillating like a free traveling wave.
Region III: After the barrier, [math]\displaystyle{ x\gt L }[/math]
[math]\displaystyle{ -\frac{\hbar^2}{2m}\frac{d^2\psi_{III}}{dx^2}=E\psi_{III} }[/math]
Since there is no incoming wave from the far right, the transmitted wave is
[math]\displaystyle{ \psi_{III}(x)=Fe^{ikx} }[/math].
The constants are
[math]\displaystyle{ k=\frac{\sqrt{2mE}}{\hbar} }[/math]
and
[math]\displaystyle{ \kappa=\frac{\sqrt{2m(V_0-E)}}{\hbar} }[/math].
The quantity [math]\displaystyle{ k }[/math] describes the oscillating wave outside the barrier. The quantity [math]\displaystyle{ \kappa }[/math] describes the exponential decay inside the barrier.
Boundary Conditions
The wavefunction and its derivative must match at both edges of the barrier.
At [math]\displaystyle{ x=0 }[/math]:
[math]\displaystyle{ A+B=C+D }[/math]
[math]\displaystyle{ ik(A-B)=-\kappa C+\kappa D }[/math]
At [math]\displaystyle{ x=L }[/math]:
[math]\displaystyle{ Ce^{-\kappa L}+De^{\kappa L}=Fe^{ikL} }[/math]
[math]\displaystyle{ -\kappa Ce^{-\kappa L}+\kappa De^{\kappa L}=ikFe^{ikL} }[/math]
These four equations can be solved for the unknown amplitudes [math]\displaystyle{ B }[/math], [math]\displaystyle{ C }[/math], [math]\displaystyle{ D }[/math], and [math]\displaystyle{ F }[/math] in terms of the incoming amplitude [math]\displaystyle{ A }[/math].
Tunneling Probability
The tunneling probability is also called the transmission coefficient. It is written as
[math]\displaystyle{ T=\frac{\text{transmitted intensity}}{\text{incident intensity}} }[/math].
Since intensity is proportional to the squared amplitude,
[math]\displaystyle{ T=\frac{|F|^2}{|A|^2} }[/math].
For a rectangular barrier with [math]\displaystyle{ E\lt V_0 }[/math], the exact transmission coefficient is
[math]\displaystyle{ T=\left[1+\frac{V_0^2\sinh^2(\kappa L)}{4E(V_0-E)}\right]^{-1} }[/math].
For a thick or high barrier, where [math]\displaystyle{ \kappa L }[/math] is large, a useful approximation is
[math]\displaystyle{ T\approx 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right)e^{-2\kappa L} }[/math].
This approximation shows the most important idea:
[math]\displaystyle{ T }[/math] decreases exponentially as the barrier becomes wider or harder to penetrate.
Since the exponential term is [math]\displaystyle{ e^{-2\kappa L} }[/math], even a small increase in barrier width can cause a huge decrease in tunneling probability.
Computational Model
The model below is an original GlowScript/Trinket visualization of quantum tunneling through a rectangular potential barrier. It lets the user change the particle energy [math]\displaystyle{ E }[/math], barrier height [math]\displaystyle{ V_0 }[/math], and barrier width [math]\displaystyle{ L }[/math]. The model shows the oscillating wave before the barrier, the decaying wave inside the barrier, and the smaller transmitted wave after the barrier.
Original GlowScript/Trinket model: Quantum Tunneling through a Rectangular Potential Barrier
<iframe src="https://trinket.io/embed/glowscript/caf10538764a?start=result"
width="100%"
height="500"
frameborder="0"
marginwidth="0"
marginheight="0"
allowfullscreen>
</iframe>
If the embedded model does not load inside the wiki, open the linked Trinket model above in a new tab. This model was created for this page and is meant to show the main qualitative idea: the wavefunction does not instantly become zero inside the barrier. It decays, and the amount left on the far side depends strongly on the barrier height, barrier width, and particle energy.
The PhET quantum tunneling simulation below is also useful because it lets users compare plane waves and wave packets for different potential shapes.
<iframe src="https://phet.colorado.edu/sims/cheerpj/quantum-tunneling/latest/quantum-tunneling.html?simulation=quantum-tunneling"
width="800"
height="600"
frameborder="0"
marginwidth="0"
marginheight="0"
allowfullscreen>
</iframe>
When using the simulations, try the following:
- Set the particle energy below the barrier height.
- Increase the barrier width and observe how the transmitted wave decreases.
- Increase the barrier height and observe how the transmitted wave decreases.
- Compare a plane wave with a wave packet.
Common Exam Mistakes
- Thinking tunneling is classically allowed. If [math]\displaystyle{ E\lt V_0 }[/math], classical mechanics predicts no transmission. Tunneling is a quantum effect.
- Forgetting that the probability drops exponentially. The factor [math]\displaystyle{ e^{-2\kappa L} }[/math] means that even a small increase in barrier width can make tunneling much less likely.
- Mixing up [math]\displaystyle{ k }[/math] and [math]\displaystyle{ \kappa }[/math]. The symbol [math]\displaystyle{ k }[/math] describes the oscillating wave outside the barrier. The symbol [math]\displaystyle{ \kappa }[/math] describes the decaying wave inside the barrier.
- Saying the particle climbs over the barrier. In tunneling, the particle is not following a classical path over the barrier. The wavefunction extends through the barrier.
- Ignoring mass. Since [math]\displaystyle{ \kappa }[/math] depends on mass, heavier particles tunnel much less easily than lighter particles.
Examples
Simple
An electron has energy [math]\displaystyle{ 5.0\text{ eV} }[/math]. What is the approximate probability that it will tunnel through a potential barrier of height [math]\displaystyle{ 10.0\text{ eV} }[/math] and width [math]\displaystyle{ 1.00\text{ nm} }[/math]?
Solution
Given:
- [math]\displaystyle{ E=5.0\text{ eV} }[/math]
- [math]\displaystyle{ V_0=10.0\text{ eV} }[/math]
- [math]\displaystyle{ L=1.00\text{ nm}=1.00\times10^{-9}\text{ m} }[/math]
- [math]\displaystyle{ m_e=9.11\times10^{-31}\text{ kg} }[/math]
- [math]\displaystyle{ \hbar=1.055\times10^{-34}\text{ J}\cdot\text{s} }[/math]
- [math]\displaystyle{ 1\text{ eV}=1.602\times10^{-19}\text{ J} }[/math]
First calculate [math]\displaystyle{ \kappa }[/math]:
[math]\displaystyle{ \kappa=\frac{\sqrt{2m(V_0-E)}}{\hbar} }[/math]
[math]\displaystyle{ \kappa= \frac{\sqrt{2(9.11\times10^{-31})(5.0)(1.602\times10^{-19})}}{1.055\times10^{-34}} }[/math]
[math]\displaystyle{ \kappa\approx1.15\times10^{10}\text{ m}^{-1} }[/math]
Now use the approximation:
[math]\displaystyle{ T\approx 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right)e^{-2\kappa L} }[/math]
[math]\displaystyle{ T\approx 16\left(\frac{5.0}{10.0}\right)\left(1-\frac{5.0}{10.0}\right)e^{-2(1.15\times10^{10})(1.00\times10^{-9})} }[/math]
[math]\displaystyle{ T\approx4.5\times10^{-10} }[/math]
So the tunneling probability is approximately
[math]\displaystyle{ \boxed{4.5\times10^{-10}} }[/math].
This is a very small probability, but it is not zero.
Middling
An electron with energy [math]\displaystyle{ 4.0\text{ eV} }[/math] approaches a rectangular barrier of height [math]\displaystyle{ 8.0\text{ eV} }[/math]. Compare the tunneling probability when the barrier width is [math]\displaystyle{ 0.50\text{ nm} }[/math] and when the barrier width is [math]\displaystyle{ 1.00\text{ nm} }[/math].
Solution
Given:
- [math]\displaystyle{ E=4.0\text{ eV} }[/math]
- [math]\displaystyle{ V_0=8.0\text{ eV} }[/math]
- [math]\displaystyle{ V_0-E=4.0\text{ eV} }[/math]
Calculate [math]\displaystyle{ \kappa }[/math]:
[math]\displaystyle{ \kappa=\frac{\sqrt{2m(V_0-E)}}{\hbar} }[/math]
[math]\displaystyle{ \kappa\approx1.02\times10^{10}\text{ m}^{-1} }[/math]
The approximate tunneling probability is
[math]\displaystyle{ T\approx 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right)e^{-2\kappa L} }[/math].
Since [math]\displaystyle{ E/V_0=4.0/8.0=0.50 }[/math],
[math]\displaystyle{ 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right)=16(0.50)(0.50)=4 }[/math].
For [math]\displaystyle{ L=0.50\text{ nm} }[/math]:
[math]\displaystyle{ T\approx 4e^{-2(1.02\times10^{10})(0.50\times10^{-9})} }[/math]
[math]\displaystyle{ T\approx1.4\times10^{-4} }[/math].
For [math]\displaystyle{ L=1.00\text{ nm} }[/math]:
[math]\displaystyle{ T\approx 4e^{-2(1.02\times10^{10})(1.00\times10^{-9})} }[/math]
[math]\displaystyle{ T\approx5.0\times10^{-9} }[/math].
Doubling the width does not merely cut the probability in half. It reduces the tunneling probability by many orders of magnitude because the width appears inside an exponential.
Difficult
An electron with energy [math]\displaystyle{ 2.0\text{ eV} }[/math] approaches a rectangular barrier of height [math]\displaystyle{ 5.0\text{ eV} }[/math]. What barrier width gives an approximate tunneling probability of [math]\displaystyle{ 1.0\times10^{-6} }[/math]?
Solution
Given:
- [math]\displaystyle{ E=2.0\text{ eV} }[/math]
- [math]\displaystyle{ V_0=5.0\text{ eV} }[/math]
- [math]\displaystyle{ T=1.0\times10^{-6} }[/math]
- [math]\displaystyle{ V_0-E=3.0\text{ eV} }[/math]
Start with the approximate tunneling equation:
[math]\displaystyle{ T\approx 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right)e^{-2\kappa L} }[/math].
Calculate the coefficient:
[math]\displaystyle{ 16\frac{E}{V_0}\left(1-\frac{E}{V_0}\right) = 16\left(\frac{2.0}{5.0}\right)\left(1-\frac{2.0}{5.0}\right) }[/math]
[math]\displaystyle{ =16(0.40)(0.60)=3.84 }[/math].
So
[math]\displaystyle{ 1.0\times10^{-6}=3.84e^{-2\kappa L} }[/math].
Divide both sides by [math]\displaystyle{ 3.84 }[/math]:
[math]\displaystyle{ e^{-2\kappa L}=2.60\times10^{-7} }[/math].
Take the natural logarithm of both sides:
[math]\displaystyle{ -2\kappa L=\ln(2.60\times10^{-7}) }[/math]
[math]\displaystyle{ -2\kappa L\approx -15.16 }[/math].
Now solve for [math]\displaystyle{ L }[/math]:
[math]\displaystyle{ L=\frac{15.16}{2\kappa} }[/math].
Calculate [math]\displaystyle{ \kappa }[/math]:
[math]\displaystyle{ \kappa=\frac{\sqrt{2m(V_0-E)}}{\hbar} }[/math]
[math]\displaystyle{ \kappa= \frac{\sqrt{2(9.11\times10^{-31})(3.0)(1.602\times10^{-19})}}{1.055\times10^{-34}} }[/math]
[math]\displaystyle{ \kappa\approx8.87\times10^9\text{ m}^{-1} }[/math].
Therefore,
[math]\displaystyle{ L=\frac{15.16}{2(8.87\times10^9)} }[/math]
[math]\displaystyle{ L\approx8.54\times10^{-10}\text{ m} }[/math].
So the barrier width is approximately
[math]\displaystyle{ \boxed{0.854\text{ nm}} }[/math].
Applications
Nuclear Fusion in Stars
Quantum tunneling helps explain how nuclear fusion can occur inside stars. Protons repel each other because they are positively charged. Classically, many protons in a star's core would not have enough energy to overcome this electric repulsion. Quantum tunneling gives them a small probability of getting close enough for the strong nuclear force to bind them together.
Alpha Decay
Alpha decay occurs when an unstable nucleus emits an alpha particle. The alpha particle is trapped inside the nucleus by a potential barrier. Classically, it may not have enough energy to escape. Quantum mechanically, it can tunnel through the barrier and leave the nucleus.
Scanning Tunneling Microscopy
A scanning tunneling microscope, or STM, uses tunneling current to image surfaces at the atomic scale. When a sharp conducting tip is brought very close to a surface, electrons can tunnel between the tip and the surface. The tunneling current is extremely sensitive to distance, which allows the STM to detect tiny changes in surface height.
Semiconductor Devices
Some semiconductor devices rely on tunneling. Tunnel diodes, flash memory, and very small transistors can involve tunneling effects. As electronic devices become smaller, tunneling becomes more important because electrons interact with barriers only a few nanometers wide.
Connection to Matter and Interactions
In Matter and Interactions, energy is often used to decide what motion is possible. For a classical particle, if [math]\displaystyle{ E\lt V_0 }[/math], the particle cannot cross the barrier. Quantum tunneling is interesting because it shows where that classical energy reasoning stops being complete.
The particle still does not have enough classical energy to go over the barrier. The difference is that quantum mechanics describes the particle with a wavefunction. That wavefunction can extend into the barrier and sometimes reach the other side. So instead of asking only whether the particle has enough energy to cross, we ask what probability the wavefunction gives for finding the particle past the barrier.
Historical Background
Quantum tunneling was developed during the early years of quantum mechanics. Friedrich Hund discussed wavefunction penetration in molecular systems in the 1920s. George Gamow later applied tunneling to alpha decay, showing that quantum mechanics could explain how alpha particles escape from atomic nuclei. Tunneling soon became one of the clearest examples of how quantum mechanics differs from classical mechanics.
See Also
- Heisenberg Uncertainty Principle
- Wave-Particle Duality
- Schrodinger Equation
- Quantum Wells and Bound States
- Solution for a Single Particle in a Semi-Infinite Quantum Well
- Application of Statistics in Physics
External Resources
- Wikimedia Commons: Quantum-tunneling.svg
- PhET: Quantum Tunneling and Wave Packets
- OpenStax: Quantum Tunneling of Particles through Potential Barriers
- LibreTexts: Quantum Tunneling of Particles through Potential Barriers
- HyperPhysics: Barrier Penetration
References
- MikeRun, Quantum-tunneling.svg, Wikimedia Commons. Licensed under Creative Commons Attribution-ShareAlike 4.0 International.
- OpenStax, University Physics Volume 3, Chapter 7: Quantum Mechanics. Licensed under Creative Commons Attribution 4.0 International.
- LibreTexts, Quantum Tunneling of Particles through Potential Barriers.
- HyperPhysics, Barrier Penetration.
- PhET Interactive Simulations, Quantum Tunneling and Wave Packets.
- Original GlowScript/Trinket model by Haripratiik Arunkumar Malarkodi, Spring 2026.