Heisenberg Uncertainty Principle: Difference between revisions
No edit summary |
No edit summary |
||
| Line 9: | Line 9: | ||
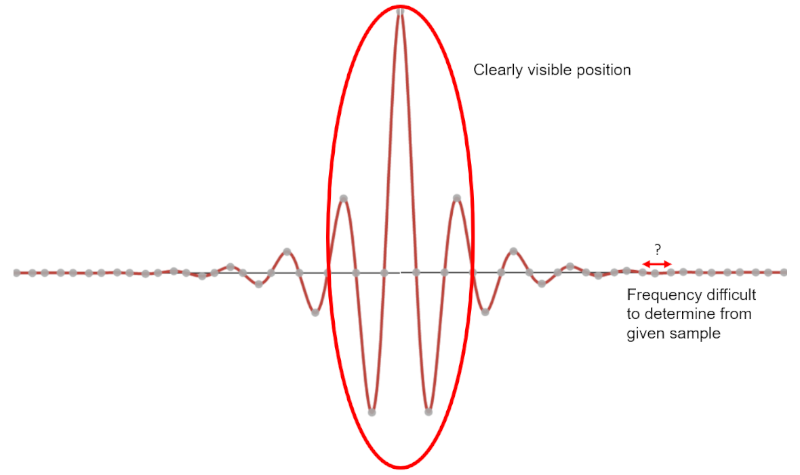
Generally speaking, uncertainty relationships are not unique to modern physics. This is a concept that occurs in classical physics as well. Consider a wave, described by some combination of sines and cosines. Any wave has an amount of uncertainty between its frequency and its position. For example, a wave with a single period would give you a lot of information about its position, but not much information about its frequency. Conversely, a wave of many periods would allow you to precisely determine its frequency, but the position of the wave would be less certain. | Generally speaking, uncertainty relationships are not unique to modern physics. This is a concept that occurs in classical physics as well. Consider a wave, described by some combination of sines and cosines. Any wave has an amount of uncertainty between its frequency and its position. For example, a wave with a single period would give you a lot of information about its position, but not much information about its frequency. Conversely, a wave of many periods would allow you to precisely determine its frequency, but the position of the wave would be less certain. | ||
[[File: | [[File:Waveimg3.png|border|caption]] | ||
===A Mathematical Model=== | ===A Mathematical Model=== | ||
Revision as of 11:43, 18 April 2022
Claimed by Bernardo Perez 4/17/22
Short Description of Topic
Heisenberg Uncertainty Principle
The Heisenberg uncertainty principle is a concept that describes uncertainty relationships between a particle's different properties, those being position and momentum, as well as energy and time.The biggest takeaway from this concept is that there is always some measure of uncertainty in at least one of any given particle's properties.
Generally speaking, uncertainty relationships are not unique to modern physics. This is a concept that occurs in classical physics as well. Consider a wave, described by some combination of sines and cosines. Any wave has an amount of uncertainty between its frequency and its position. For example, a wave with a single period would give you a lot of information about its position, but not much information about its frequency. Conversely, a wave of many periods would allow you to precisely determine its frequency, but the position of the wave would be less certain.
A Mathematical Model
The total uncertainty between a particle's position and momentum is given as [math]\displaystyle{ {\Delta p \Delta x} }[/math]. The minimum uncertainty a particle can have is [math]\displaystyle{ {\frac{{\hbar}}{2}} }[/math]. Therefore, we can establish the relationship of:
[math]\displaystyle{ {\Delta p \Delta x \gt = \frac{{\hbar}}{2}} }[/math]
Oftentimes, it is also good enough to simply approximate their general relationship as:
[math]\displaystyle{ {\Delta p \Delta x} }[/math] ~ [math]\displaystyle{ {\hbar} }[/math]
NOTE: [math]\displaystyle{ {\hbar = \frac{{h}}{2\pi}} }[/math] where [math]\displaystyle{ {h} }[/math] is Planck's constant.
This principle can also be extended to a relationship between energy and time, where:
[math]\displaystyle{ {\Delta E \Delta t \gt = \frac{{\hbar}}{2}} }[/math]
and
[math]\displaystyle{ {\Delta E \Delta t} }[/math] ~ [math]\displaystyle{ {\hbar} }[/math]
A Computational Model
How do we visualize or predict using this topic. Consider embedding some vpython code here Teach hands-on with GlowScript
Examples
Be sure to show all steps in your solution and include diagrams whenever possible
Simple
Middling
Difficult
Connectedness
- How is this topic connected to something that you are interested in?
- How is it connected to your major?
- Is there an interesting industrial application?
History
The development of the Heisenberg uncertainty principle is largely accredited to Werner Heisenberg.
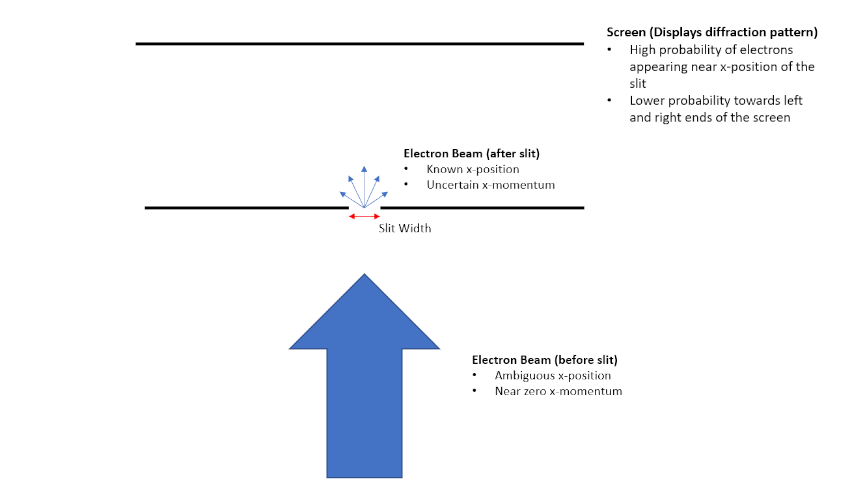
Single-slit Diffraction Experiments
Heisenberg uncertainty relationships were first physically demonstrated through single-slit diffraction experiments. The experiment is conducted by launching beam of electrons through a narrow slit. Initially, the electrons have zero momentum in the x-direction. After passing through the slit, information about an electron's x position is acquired, resulting in an increased uncertainty in the particle's x momentum. Making the slit narrower therefore increases the uncertainty of the particle's x momentum while decreasing the uncertainty of its x position, while making the slit wider has the opposite effect. This effectively demonstrates the relationship between the uncertainty of a particle's position and momentum.
See also
Are there related topics or categories in this wiki resource for the curious reader to explore? How does this topic fit into that context?
Further reading
Books, Articles or other print media on this topic
External links
Internet resources on this topic
References
Krane, K. S. (2020). Modern physics. Wiley.