Josiah Willard Gibbs: Difference between revisions
| Line 38: | Line 38: | ||
Another famous theory he developed is known as Gibbs Phase Rule. This theory applies to heterogenous systems in thermodynamic equilibrium and calculates the number of possible phases in a system. | Another famous theory he developed is known as Gibbs Phase Rule. This theory applies to heterogenous systems in thermodynamic equilibrium and succinctly calculates the number of possible phases in a system. | ||
[[File:Phase-rule-4-638.jpg]] | |||
In his goal to find simpler approaches to science, Gibbs played a pivotal role in popularizing the use of vector notation in physics. This more straight forward method replaced the more complex method of quaternions which were previously used. | |||
===Significance of Gibbs' Scientific Contributions=== | ===Significance of Gibbs' Scientific Contributions=== | ||
Revision as of 22:03, 30 November 2015
editing in process by msenderowitz3. plz don't take
Personal Life
Early Life
Josiah Willard Gibbs was born to Josiah Gibbs Sr. and Mary Anna Van Cleve on February 11, 1839 in New Haven, Connecticut. As a child he lived a relatively privileged life with his four older sisters. Education was encouraged in his family as not only was his father a professor at Yale, but also had one relative who held a position as president of Harvard and another relative who was the first president of Princeton. His classmates at The Hopkins School, a small private school in New Haven, often described him as quiet and "intellectually absorbed." His fragile pulmonary and overall health likely contributed to his introverted demeanor as it prevented him from interacting with his peers.
Later Life
Gibbs continued his pursuit of education when he matriculated to Yale when he was only 15 years old (in 1854). There, he began to pursue engineering research while also receiving numerous awards for his exceptional academic performance in Latin and Mathematics. After successfully completing his undergraduate career, Gibbs continued with graduate studies at Yale. In 1863, at the age of 19, Josiah Willard Gibbs became the first American to receive a doctorate in engineering. It is evident that his academic accomplishments were made possible through his intellect and work ethic, but it is worth noting that his health problems prevented him from serving in the Civil War and allowed him to focus on his studies.
Several years after finishing his graduate work and completing three years as a tutor at Yale, Josiah Willard Gibbs journeyed to Europe with his sisters where he attended lectures on mathematics and physics. After his three years abroad, Gibbs returned to America with a more european view of science. This was one of the reasons that despite his future scientific work, notoriety came very slowly, if at all, in the United States.
Gibbs became a pioneer in another aspect of academia when in 1871, he became the first professor of Mathematical Physics in the United States. Due to his financial security from his inheritance from his parents, he taught without pay for nine years at Yale.
During his remaining years, Josiah Willard Gibbs maintained his reclusive lifestyle. Not only did he never marry, but he also remained living in his childhood home with his older sister and her husband. Despite a few vacations to the Adirondacks and New Hampshire, Gibbs spent the rest of his life in New Haven either working at Yale or in his home. He died April 28th 1903 from acute intestinal obstruction.
Scientific _______
Early Scientific Career
Despite his intellect, Josiah Willard Gibbs did not attract much attention in the scientific community for several reasons. One of these being that American colleges at the time did not encourage research, another being academia focused more on "practical" questions rather than theoretical ones. Additionally, his writings were esoteric in a unique sense; chemists found his papers to be to mathematical, while mathematicians found them to be too scientific.
However, due to his theoretical capabilities, he was able to develop one of his most prominent achievements. By analyzing James Watt's steam engine governor and analyzing its equilibrium, he bagan to develop an equation to calculate and quantify the equilibriums of chemical processes. (This will be discussed in further detail shortly).
Scientific Contributions
As mentioned before, his brilliance in the scientific field was unfortunately not appreciated at the time however made lasting impacts on the scientific community.
Arguably, his most famous contribution was the concept of Gibbs Free Energy. In short this relates a systems energy and entropy. He believed that understanding a systems equilibrium (the maximum of a systems' entropy) was essential in understanding the system as a whole. His equations developed allow one the calculate a system's free energy, how fast the reaction will occur, its direction, and spontaneity. He was the first person to derive a differential equation relating temperature, entropy, energy, pressure and volume which can be simplified into the following equation:
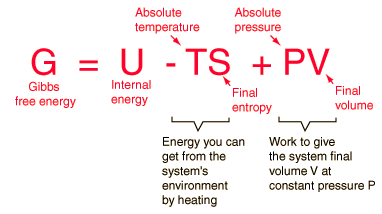
This theory can also be used to find the change in Gibbs Free Energy as shown in the following equation:
 If the numerical value for Delta G is negative, the reaction is spontaneous.
If the value is positive, the reaction is non-spontaneous.
If the value is zero, the system is in its equilibrium state.
If the numerical value for Delta G is negative, the reaction is spontaneous.
If the value is positive, the reaction is non-spontaneous.
If the value is zero, the system is in its equilibrium state.
Another famous theory he developed is known as Gibbs Phase Rule. This theory applies to heterogenous systems in thermodynamic equilibrium and succinctly calculates the number of possible phases in a system.

In his goal to find simpler approaches to science, Gibbs played a pivotal role in popularizing the use of vector notation in physics. This more straight forward method replaced the more complex method of quaternions which were previously used.
Significance of Gibbs' Scientific Contributions
Gibbs' contributions forever changed the way the world looks at and studies science and laid the groundwork for many future scientists' discoveries/theories.
Due to his works in thermodynamics, much of physical chemistry was changed into a deductive science rather than solely an empirical one.
-realized that some of his findings had already been discovered before, but aimed to make a more simple approach -although well liked by friends, US scientists didn’t give him much attention as American Science was more focused on practical questions than his theoretical work -praiseworthy that he didn’t really care that the scientific did not appreciate his works. He knew the significance of his findings and “was content to let posterity appraise him” -his works paved the way for many of Albert Einsteins later discoveries -from 1876-1878, while working on principles of thermodynamics, discovered concept of chemical potential
patented design for railway break