Charge Transfer
claimed by Kate Blake
If a charged conductor comes in contact, or is in close enough proximity, with another conductor, it is possible to transfer this charge to the second conductor. This process is called charge transfer. However, the Law of Conservation of Charge states that charges cannot be created or destroyed. Charge cannot be created, the presence of a negative charge is merely the effect of an object gaining electrons from another material. Since charge cannot be created or destroyed, and is just the transfer of electrons between materials, the magnitude of the charge transfer between two objects will be equivalent. For however much the charge of one object increases during charge transfer, the other must decrease the same amount. There are multiple ways that charge can be transferred such as through direct contact (ie friction), induction, and conduction.
The Main Idea
Insulators vs Conductors

In an insulator, electrons are bounded tightly to atoms, which prevents charged particles from moving through the material. If charge is transferred to an insulator at a given location, the charge will remain at the location that the transfer occurred. Examples of insulators include rubber and air.
Within conductors, on the other hand, electrons are able to flow freely from particle to particle. When charge is transferred to a conductor, the charge is distributed evenly across the surface of the object via electron movement. The electrons will be distributed until the repelling force between the excess electrons is minimized. This is the main difference between insulators and conductors: insulators do not have mobile charged particles whereas conductors have mobile charged particles that allow for charge transfer through the free movement of electrons. Examples of conductors include metals and salt water.
Charge by Friction
Certain objects and materials have a greater attraction to electrons than others. For example, rubber is highly attracted to electrons, whereas fur or hair has a lower attraction. Therefore, if you take a balloon and rub it on your head, electrons previously in the atoms of the hair will be pulled to the atoms of the rubber balloon. This creates an electron imbalance which makes the balloon negatively charged and the hair positively charged. This creates the effect where the hair will stand up and be pulled towards the balloon since the opposing charges make the two objects attracted. Likewise, two charged balloons will repel each other. Another example of this is rubbing a glass rod with silk. The glass rod will become positively charged and the silk will become negatively charged; this means that electrons were transferred from the glass rod to the silk, since protons are not removed from the nuclei. Rubbing two objects together is not necessary for charge transfer, but because rubbing creates more points of contact between two objects, it facilitates charge transfer.
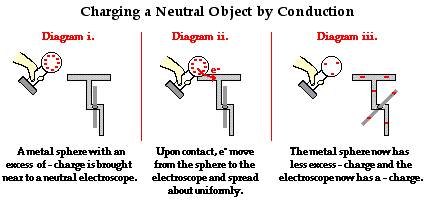
Transfer Charges by Conduction
As shown in the previous section, electrons move from one object to another through points of contact; this is especially true among metals. Charging by conduction requires the contact of a charged metal, positive or negative, to a neutral metal. If a negatively charged object touches the neutral metal, the excess electrons will flow through the neutral object. Since electrons repel each other and the negatively charged metal has a buildup of electrons, a certain number of excess electrons will flow out and spread across the neutral object when given an outlet in the form of the neutral metal. This process leaves both metals negatively charged. The same process occurs with positively charged objects touching neutral metals.
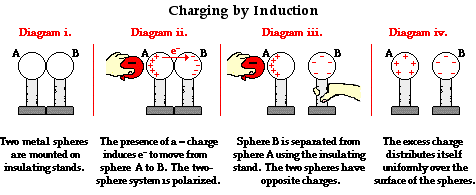
Transfer Charges by Induction
Unlike the transfer of charges by conduction, objects can transfer charges by induction without making contact. When an object is charged, it has an electric field. This electric field will repel or attract electrons in another object. This electron movement is called transfer of charges by induction. A neutral object can be charged by another charged object through a process called polarization. This is when electrons in the object are repelled or attracted to one side of the object by the charged second object. For example, if a negatively charged sphere is placed near a neutral sphere, the electrons in the neutral sphere will be repelled by the charged sphere. The neutral sphere is now polarized, with one side of it being negatively charged and the other side being positively charged. The negatively charged side of the sphere can be removed through grounding or with a conductor. Once removed, the originally neutral sphere will now be positively charged. Another example of induction is the balloon and black pepper experiment. A balloon can be given a negative charge by rubbing it on hair. When the balloon is placed near grounded black pepper, the black pepper particles will be polarized so that they become positively charged on top and will be attracted to the negatively charged balloon.
A Mathematical Model
When a conductor with charge [math]\displaystyle{ \bigl( Q_{1} \bigl) }[/math] comes into contact with another conductor with charge [math]\displaystyle{ \bigl( Q_{2} \bigl) }[/math], The charge will evenly distribute itself over both conductors.
This can be represented through the equation:
([math]\displaystyle{ \bigl( Q_{1} \bigl) }[/math] + [math]\displaystyle{ \bigl( Q_{2} \bigl) }[/math]) = [math]\displaystyle{ \bigl( Q_{total} \bigl) }[/math] , where [math]\displaystyle{ \bigl( Q_{total} \bigl) }[/math] is evenly distributed along the area of both conductors.
A Computational Model
This website is a great model for charge transfer. It shows charge transfer between a sweater and a balloon to demonstrate static electricity. Try using it without the charges showing and guess where they will go.
Examples
Simple
A student said, "When you touch a charged piece of metal, the metal is no longer charged; all the charge on the metal is neutralized." As a practical matter, this is nearly correct, but it isn't exactly right. What's wrong with saying that all the charge on the metal is neutralized? (Q15 from page 579 of Matter and Interactions)
Touching a charged piece of metal actually causes charge transfer. Electrons move from whichever item is more positively charged, or more lacking in electrons. What the student said is not entirely correct because the metal loses it's charge by the movement of electrons (negative charges).
Middling
You take two invisible tapes of some unknown brand, which we will call U and L, stick them together, and discharge the pair before pulling them apart and hanging them from the edge of your desk. When you bring an uncharged plastic pen within 10 cm of either the U tape or the L tape you see a slight attraction. Next you rub the pen through your hair, which is known to charge the pen negatively. Now you find that if you bring the charged pen within 8 cm of the L tape you see a slight repulsion, and if you bring the pen within 12 cm of the U tape you see a slight attraction. Briefly explain all of your observations. (Q21 from page 580 of Matter and Interactions)
If a plastic pen is rubbed through your hair, it gets negatively charged. There is a slight repulsion with L-tape and negatively charged plastic pen. So the charge on the L-tape should be negative. There is a slight attraction with U-tape and negatively charged plastic pen. So the charge on the U-tape should be positive.
Difficult
You run your finger along the slick side of a positively charged tape, and then observe that the tape is no longer attracted to your hand. Which of the following are not plausible explanations for this observation? Check all that apply. (1) Sodium ions (Na+) from the salt water on your skin move onto the tape, leaving the tape with a zero (or very small) net charge. (2) Electrons from the mobile electron sea in your hand move onto the tape, leaving the tape with a zero (or very small) net charge. (3) Chloride ions (Cl-) from the salt water on your skin move onto the tape, leaving the tape with a zero (or very small) net charge. (4) Protons are pulled out of the nuclei of atoms in the tape and move onto your finger. (P61 from page 585 of Matter and Interactions)
When Sodium ions (Na+) from the salt water on your skin move onto the tape, they provide additional positive charge to the already positive charged tape. So 1) is not a plausible explanation.
Human hand is considered neutral so no electrons travel from the hand to the positively charged tape so the net charge remains the same as before. So 2) is not a plausible explanation.
Chlorine particles are negative and they neutralize the positive tape. So tape then has no net charge and can't attract the hand. So 3) is a plausible explanation.
There is no chance of protons getting pulled out of the atoms nucleus as they are bound very strongly by the strong nuclear force. So 4) is not a plausible explanation.
Connectedness
A very simple example of charge transfer exists within most homes:
- The Carpet Shock
- The age old trick where you rub your feet along a carpet and then get shocked by a door knob involves charge transfer. When you rub your feet along the carpet, your feet pick up a slightly negative charge. When your hand then moves to the door knob, which is usually metal and therefore a conductor, the electrons want to spread evenly through your body and the door knob, creating a slight shock when the charge transfers.
History
The first evidence of charge transfer was discovered by Sir William Watson in 1746 while working with the Leyden jar. He later discovered that Benjamin Franklin had produced similar results in his own experiments, and the two began working closely together. Though the two men were the first to propose the principle, it was not until 1843 that Michael Faraday provided what was considered conclusive proof of this phenomenon.
See also
Further Reading
External Links
More on charge transfer.
More on charging by conduction.
This video talks about methods of charge transfer and using an electroscope to measure charge.
References
Internet resources on this topic:
http://www.physicsclassroom.com/class/estatics/Lesson-1/Conductors-and-Insulators
http://www.physicsclassroom.com/class/estatics/Lesson-1/Charge-Interactions
Chabay, Ruth W., Bruce Sherwood. Matter and Interactions, Volume II: Electric and Magnetic Interactions, 4th Edition. Wiley, 19/2015. VitalBook file.
Purrington, Robert D. (1997). Physics in the Nineteenth Century. Rutgers University Press.